Surgery in AMD
Surgery in AMD
Authors:
Angelina Meireles, MD
Hospital Santo António-CHP - Porto, Portugal
Bernardete Pessoa, MD
Hospital Santo António-CHP - Porto, Portugal
Natália Ferreira, MD
Hospital Santo António-CHP - Porto, Portugal
Rui Martinho, MD
Hospital Lusíadas Porto - Porto, Portugal
Updated/reviewed by the authors, July 2017.
Introduction
In recent years, new treatment modalities such as photodynamic therapy and intravitreal anti-VEGF injections have been added to the treatment armamentarium for age-related macular degeneration (AMD).
Prior to the introduction of these therapies, only laser photocoagulation had been shown in a large randomized controlled trial to confer a statistically significant benefit in the treatment of subfoveal choroidal neovascularization (CNV) secondary to AMD regarding to long-term visual acuity when compared to the natural history of the condition(1-3).
Unfortunately, the Macular Photocoagulation Study also showed that laser photocoagulation was associated with immediate average reduction of visual acuity, with benefits over no treatment apparent only six months after treatment(1-3).
In fact, recovery of good vision rarely occurred in these patients.
As a result of the limited treatment options, alternative therapies, such as submacular surgery for CNV removal, were pursued with limited or no success.
Meanwhile, a different management paradigm for AMD was established with macular translocation.
However, because of its higher risk of complications, its popularity has waned with the wider availability of photodynamic therapy and the introduction of intravitreal anti-VEGF agents.
Nonetheless, they remain potentially useful treatment options, even if their role in the management of AMD is neither established nor consensual.
In the meantime, vitreoretinal surgery and vitrectomy have had important developments, mostly due to advances in sutureless transconjunctival vitrectomy, use of dyes, tamponade agents and new equipments.
Novel surgical approaches for AMD are under scrutiny: the association of submacular surgery with pigment cell transplantation, the use of adjuncts, such as recombinant tissue plasminogen activator (r-TPA) for subretinal hemorrhages displacement combined with anti-VEGF treatment.
Recently, a new insight involving the vitreoretinal surface has been proposed for the pathophysiologic mechanisms underlying the development of CNV in AMD.
Accordingly, posterior vitreomacular adhesion may be another risk factor in a subpopulation of patients with wet AMD, opening another path for a surgical approach in the treatment algorithm for this condition(4-7).
Future studies are needed to define the role of vitreoretinal surgery in such cases.
Macular translocation
The first experiments on retinal relocation were conducted and reported in the beginning of the 80’s.
Their aim was to study the anatomic dependency of the foveal retina on foveal retinal pigment epithelium (RPE) and choroid.
The assumption that visual function could be preserved with foveal displacement has originated from cases of ectopic macula from retinal traction or after surgery in patients with retinopathy of prematurity and retinal detachments with giant tears(8).
They ended up showing the feasibility of rotating the macula around the optic disc with reattachment of the fovea in animal eyes(9).
One might say that this was the starting point for the idea of rotating the macula of eyes with subfoveal CNV to a new area of underlying RPE-Bruch’s membrane-choriocapillaris complex – macular translocation – as a treatment for the condition.
Although the exact pathogenesis of CNV secondary to AMD is not known, the natural history of this condition is a progressive loss of central vision over time.
The initial retinal dysfunction responsible for impaired vision in eyes with subfoveal CNV may be attributable to factors such as subretinal fluid, subretinal hemorrhage and retinal edema.
When fibrous proliferation and degeneration of the overlying photoreceptors occur during the later stages of the disease, the visual loss becomes irreversible.
The rationale of macular translocation is that moving the neurosensory retina of the fovea in one eye with recent-onset subfoveal CNV to a new location before the occurrence of permanent retinal damage, may allow it to recover or to maintain its visual function over a healthier bed of RPE-Bruch’s membrane-choriocapillaris complex.
In addition, relocating the fovea to an area outside the CNV allows the ablation of the later, by laser photocoagulation without destroying the fovea, in an attempt to preserve central vision.
On the other hand, some surgeons have combined macular translocation with CNV removal, allowing the fovea to be relocated to an area outside the RPE defect created during submacular surgery.
Several different surgical techniques for macular translocation have been described and were currently used.
These techniques produce different degrees of postoperative foveal displacement, and can broadly be classified into two categories:
Full macular translocation, and limited macular translocation.
Full macular translocation
After developing their surgical techniques in rabbit eyes(9), Machemer and Steinhorst became, in 1993, the first surgeons to demonstrate the feasibility of macular translocation in humans(10,11).
Their technique involved lensectomy, complete vitrectomy, planned total retinal detachment by transretinal infusion of fluid under the retina, 360º peripheral circumferential retinotomy, rotation of the retina around the optic disc, and reattachment of the retina with silicone oil tamponade.
Besides allowing retinal rotation to occur, the retinotomy also made way to the subretinal space for syncronous blood and CNV removal.
Since then, a number of investigators have subsequently modified this technique(12-15).
Corrective extraocular muscle surgery for globe counter-rotation, due to frequent postoperative cyclovertical diplopia or awareness of a tilted image, may be done during the primary surgery or at a later stage(13,16).
While some surgeons have found the results of macular translocation encouraging(13,17-20), others found the surgery unpredictable(21,22).
In a series of 50 consecutive eyes with subfoveal CNV from AMD that underwent full macular translocation followed-up for a median period of 21 months (12-36 months), Pertile and Claes reported an improvement of ≥ 2 Snellen lines in 66%, while 28% remained stable (±1 line) and 6% decreased by ≥ 2 lines(17).
Van Romunde et al. published long-term results of full macular translocation, in a retrospective study, of 158 patients with subfoveal CNV from AMD followed-up for a mean period of 45 months (12-108 months). A clinically and statistically significant improvement of best-corrected visual acuity (BCVA) was detected and sustained for up to 5 years after full macular translocation. They found that 45% had a 3-line or more gain at last visit. In a subgroup of 56 patients followed up for 5 years or more, there was a gain of 1.5 lines compared with preoperative BCVA(19).
In a series of 40 consecutive patients with subfoveal CNV from AMD, who undergone the mentioned surgical technique and with a mean follow-up period of 37.6 months (12.4-67.4 months), Chen et al. reported at the last observation that 68% achieved a BCVA of 6/60 or better, 43% achieved 6/30 and 15% 6/12 or better. A 3-line gain in BCVA was obtained in 30% at 1 year and in 25% at the most recent follow-up. However, in 25% of patients achieving 6/60 or better at 1 year, BCVA declined by greater than 2 lines primarily as a result of recurrent CNV(20). In another prospective, interventional, consecutive non-comparative case series of 61 AMD patients who underwent the same procedure and followed-up for 12 months, the visual acuity improved by ≥ 1 Snellen lines in 52%(23).
Toth et al. also showed improvements in distance and near visual acuity, contrast sensitivity and reading speed in a series of 25 consecutive AMD patientss(24).
In addition to exceptional surgical technique, avoiding intra and postoperative complications, the key for success after macular translocation seems to be patient selection(25,26).
If this procedure is performed on a patient without viable foveal photoreceptors, there is no chance for visual improvement.
Still, the main drawback of macular translocation lies on its high rate of complications. Rhegmatogenous retinal detachment, with or without proliferative vitreoretinopathy development, is the most common serious complication of macular translocation. Rates up to 19% have been reported(27).
Persistent or recurrent subfoveal CNV has been described in up to 30% of patients undergoing this procedure(19,27). Almost all reported cases of recurrent CNV occurred at the edge of the RPE defect created by the CNV excision. In order to reduce the recurrence of CNV, the fovea needs to be reconstructed by using healthier RPE such as those derived from stem cells (28). Chen et al. recommend close monitoring of the edge of excision site, clinically and with SD-OCT, to detect early signs of CNV. Prompt initiation of a course of anti-VEGF therapy can maintain visual acuity gained following translocation(20).With a longer follow-up, cystoid macular edema (CME) and subfoveal RPE atrophy may be limiting factors for improved postoperative visual acuity. Terasaki and colleagues found a 70% incidence rate of CME(29). Chen et al. described CME in 38 of patients (95%), at some stage postoperatively(20).
In a long-term follow-up, Aisenbrey et al. (14 to 79 months – mean 38) and von Romunde et al. (12 to 108 months – mean 45) reported 28% versus 47% of patients with subfoveal atrophy by the third and fifth year postoperatively respectively(18,19).
In figures 1 and 2, we present two anecdotal cases in which full macular translocation was performed. They presented with extensive submacular hemorrhages that were considered inadequate for anti-VEGF therapy. Visual rehabilitation was achieved, enabling the patient’s independence for daily activities.
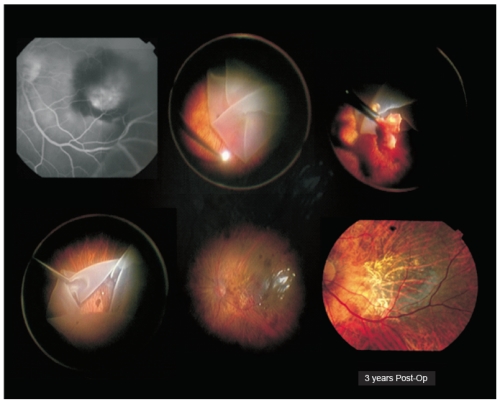
Figure 1. Case I – Patient with severe visual acuity loss in his left eye 3 days before admission (right eye was lost to advanced AMD). He presented with a submacular hemorrhage complicating wet AMD. Full macular translocation was performed (António Travassos – Coimbra Surgical Centre, Coimbra, Portugal). Three years after surgery, left eye visual acuity was 20/200.
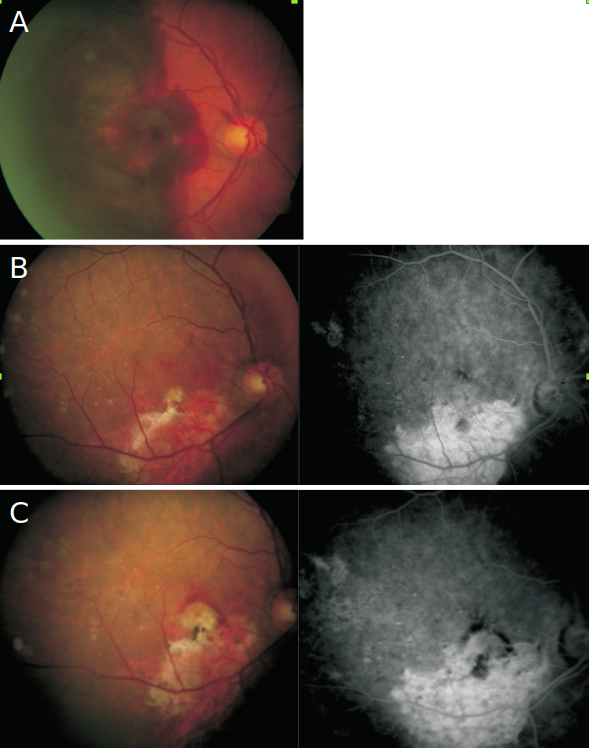
Figure 2. Case II – A 85-year-old man with an extensive submacular hemorrhage associated with subfoveal CNV (A). Four months after a full macular translocation (Claus Eckardt-Staedtische Kliniken Frankfurt A.M., Germany) his visual acuity was 20/80 (B). A recurrent subfoveal CNV developed after the first year of follow-up, and the patient was started with intravitreal ranibizumab injections. After 4 treatments, a visual acuity of 20/63 remained stable (C).
Limited macular translocation
In an effort to overcome the major complication following macular translocation with large retinotomy, proliferative vitreoretinopathy, de Juan developed a new technique in 1998(30).
His technique involved transretinal hydrodissection using small posterior retinotomies to induce a subtotal retinal detachment, a complete vitrectomy, anterior-posterior shortening near the equator and retinal reattachment.
As no large retinal break was created, the likelihood of developing proliferative vitreoretinopathy was thought to be lower.
As more experience was gained with this surgery, additional modifications were made to the original technique.
This is essentially a five-step procedure. It starts with the placement of scleral imbricating sutures (either in the superotemporal or inferotemporal quadrants for inferior and superior translocations, respectively) for antero-posterior shortening of the eyewall, followed by a pars plana vitrectomy with posterior vitreous detachment induction.
The third step should be an internal subretinal hydrodissection through small self-sealing retinotomies for creation of a partial neurosensory retinal detachment.
After tightening the scleral imbricating sutures, a redundancy of the neurosensory retina relative to the eyewall is created that should allow the desired foveal displacement, after retinal reattachment with fluid-air exchange.
Limited macular translocation may be either inferior or superior, depending on the movement of the neurosensory macula relative to the underlying tissues.
The largest series of limited macular translocation by Pieramici et al., in a retrospective review, analysed the outcome of 102 consecutive eyes of 101 patients that underwent inferior limited macular translocation for AMD-related subfoveal CNV. At three and six months postoperatively, 37% and 48% of the eyes, respectively, experienced ≥ 2 Snellen lines of visual improvement(31).
After one year of follow-up, 39.5% still maintained a ≥ 2 lines of improvement, while 29.0% remained unchanged and 31.4% lost ≥ 2 lines of visual acuity(32).
Recurrent CNV developed in 34.6% of the eyes, being subfoveal in most of the cases (65%)(32).
Besides the usual risks inherent to pars plana vitrectomy, limited macular translocation is associated with additional risks similar to those of scleral buckling surgery.
As with full macular translocation, the rate of complications won its popularity.
Although conceived in an effort to reduce the rate of proliferative vitreoretinopathy development, this has remained the most common serious complication after limited macular translocation.
Rates of retinal detachment up to 17.4% have been reported(33).
Insufficient macular translocation is another important limitation of this procedure, restricting its indications to smaller CNV.
Is there a role for macular translocation in the current era of anti-VEGF therapy and combined treatments?
IIn the latest edition of Vitreous Microsurgery (5th Edition, 2011 - Lippincott Williams & Wilkins)22),
Steve Charles et al. stated again that do not believe that this procedure is indicated due to the high incidence of complications.
In the review from Eandi et al. to access the functional outcomes of macular translocation for wet-AMD was concluded that there wasn’t enough evidence in randomized trials to support the benefit of surgery(34).
Future studies should involve patients with small neovascular membranes that are non-responsive to the present medical armamentarium, and that accept the risks of surgery in an effort to improve their visual function.
This is a procedure that demands a high level of surgical skill and experience, with a rather flat learning curve. The number of complications is unacceptably high. The widely use and quite effective pharmacologic therapy has limited this surgical modality to patients with large submacular haemorrhages, or patients unresponsive to VEGF inhibitors(19).
At the present time, the very small incidence of cases that could benefit from this procedure does not allow every Ophthalmology department with vitreoretinal surgical capability to gain expertise on macular translocation.
Referral this few cases to selected surgical units with the proper human and technical resources and experience seems the best choice.
Submacular surgery
As already has been said, a variety of surgical treatments have been developed for exudative AMD disease and one of those are the different modalities of submacular surgery.
We will review these surgical approaches and their actual role in the AMD treatment.
Surgery with subfoveal neovascular membrane removal
In 1988, Juan and Machemer(35)
published the first results of vitreous surgical techniques on removal of blood or fibrous submacular complications in four AMD patients.
Countless publications of retrospective small sample size studies ensued, with no control group, describing the benefits of this technique in stabilizing the disease, albeit displaying reduced functional benefits(36-38).
The need to establish the benefits of submacular surgery in the treatment of CNV led to the Submacular Surgery Trials (SSTs). One of the aims of this study was to find out if surgery, for the different types of AMD lesions, more than vision stabilization, was able to increase vision with real impact in the life quality of these patients(39).
The pilot trial used to pre-test the research procedures of this multicentre, randomized, controlled study found no reason to prefer submacular surgery to laser photocoagulation in AMD patients with similar lesions to those displayed by study patients(40).
Several surgical techniques are described in the literature. In summary, these techniques include standard pars plana vitrectomy, with or without posterior hyaloid membrane removal; posterior retinotomy followed by infusion of subretinal saline solution or r-TPA into lesions with a large haemorrhagic component, membrane mobilization and its removal with surgical forceps, followed by eventual aspiration of fresh blood or clot, depending on the situation.
Possible intraocular haemorrhages during surgery may be controlled by increasing the intraocular pressure, either by raising the infusion bottle or using heavy perfluorocarbon liquids.
The procedure is finished with a fluid-air exchange, followed by gas buffering, maintaining the patient in the prone position until gas reabsorption(41-44).
Due to the physiopathology of this disease, it was observed in histopathological studies that inadvertent and undesired removal of the pigment epithelium often occurs during membrane removal, especially for type 1 membranes.
The irreversible absence of the pigment epithelium leads to loss or atrophy of photoreceptors and choriocapillaries, inducing an unfavourable visual recovery(45).
The percentage of removed epithelium is variable but may reach significantly high values, as observed in the SSTs, where the pigment epithelium was involved in 84% of removed membranes(46). ther complications like regmatogeneous retinal detachment were also observed, with higher incidence in eyes with larger hemorrhagic lesions and relatively poor visual acuity(47).
Therefore, both the functional results and the impact on life quality observed for the various subgroups considered in the SSTs, compared to natural disease progression, led the authors not to recommend submacular surgery as a treatment option(48-51).
Retinal pigment epithelium transplants
As previously referred, the poor results achieved with surgical removal of subfoveal membranes, largely due to resulting atrophy or rupture of the photoreceptor-RPE complex, led some groups to combine neovascular membrane removal with simultaneous autologous transplant of iris or RPE, in order to restore normal subretinal conditions(52-54).
The surgical maneuvers were similar to those used for subfoveal CNV removal with additional aspiration and subsequent pigment epithelial cell transplant procedures.
Although good tolerability has been demonstrated for pigment cells in the subretinal space, as well as absence of membrane recurrence, the poor functional results obtained in most studies have been attributed to several factors, including lack of differentiation in pigment cell reproliferation, failure in adhesion to Bruch’s membrane and failure to form a regular pigment epithelial cell monolayer(52-56).
Also according to other authors, the behavior of transplanted cells depends essentially on the type of environment found at the seeding location(57).
Meanwhile, Peyman et al. described a technique in which an autologous pedicle graft or homologous RPE cells and Bruch’s membrane replaced the submacular RPE(58). At mid-90s, Algvere et al. transplanted fetal RPE as a monolayer patch or in a suspension form(59,60).
LLater on, Aylward et al., from Moorfields Eye Hospital, were the first team to describe the concept of total patch translocation of the RPE-choroid complex to the subfoveal area(61). The patches collected in the macular area near the RPE lesion were small and even so they survived in the subfoveal space (assessed by OCT, FA and ICG angiography); after 5 years of follow-up, none of the nine transplanted patients displayed any function, what might be explained by chronic photoreceptor apoptosis(62).
In Rotterdam, Van Meurs et al., described a modification to this technique in which a healthier full-thickness patch of RPE-choroid of approximately 1.5x2 mm is harvested from within a circular zone isolated by heavy diathermia in the superior midperiphery where cells were thought to be less damaged(63). he group reported long-term results (up to 7 years) of at least 20/200 final BCVA in 15% of patients and the main complications were recurrence of CNV (10%) and proliferative vitreoretinopathy (10%)(64).
Subsequently, other groups have been publishing small case series presenting not only expressive and sustained improvement in visual acuity and reading ability, but also recovery of central fixation, evidence for grafts revascularization (also demonstrated histologically in animals experiments) and a normal autofluorescence over the patch in patients with exudative or dry AMD(63-72).
According to Parolini et al., those eyes whose vision is not seriously impaired (equal or higher than 20/200) and with a relatively well-preserved retinal layers, especially the external limiting membrane, are the ones with better outcomes. In this series, long-lasting beneficial effect of the graft for the function of the retina with improvement of vision was observed in most cases(73). Another critical issue for a successful outcome is the blood supply to the graft. Cereda et al. reported partial revascularization of the patch with choroidal vessels running parallel to one another unlike the radially choroidal vascular system in the macula area. In some cases localized feeder vessels, usually at the graft margin, were seen. These vessels are probably a large choroidal artery arising from the connection between the patch and the original choroid or choriocapillaris(74,75).
During CNV removal, choroidal structure should be left unharmed and the dimension of the graft must be large enough to connect to an area of preserved choroid. Revascularisation is a progressive event and time to completion is variable (over 1 month). Before revascularisation, patch survival may be accomplished through plasmatic diffusion, as in skin transplantation(76).
RPE-choroid transplant offers an interesting alternative to macular translocation, especially in patients with good vision in their fellow eye. This surgery is challenging and not free from intraoperative and postoperative complications. Improvements in microsurgical techniques can lead to less traumatic CNV removal and insertion of the RPE-choroid graft. This is crucial for promising functional results, as well as an adequate patient selection.
Management of submacular haemorrhage
In addition to the poor prognosis, exudative forms of AMD evolved into a major submacular haemorrhage that are difficult to diagnose (concerning membrane location and extension) and to treat(77,78).
n the era of antiangiogenic agents, this is the most frequent form of AMD for which surgery is indicated, aiming to avoid further blood damages (mechanical, metabolic and toxic) to the photoreceptor-RPE complex(41,79,80).
As previously referred, blood was initially removed by aspiration or mechanically extracted.
In 1991, Lewis et al. were the first researchers to report the fibrinolysis properties of r-TPA, which made this agent useful in removing blood clots(42-44,81-84).
In order to avoid surgical manipulation of the macular retina, Herriot described a procedure involving displacement of submacular blood by intravitreal injection of r-TPA and gas, followed by prone position, for the first time in 1997.(84)
Blood is normally displaced temporally or infero-temporally, with a significant increase in visual acuity occurring immediately after the aforementioned procedure, as described in countless published outcomes.
Some authors have pointed out the duration of hemorrhage (the shorter the better) as the main predictive factor for the aforementioned improvement(85-92).
On the other hand, the usefulness of intravitreal r-TPA as an adjuvant to this technique has been questioned, not only because r-TPA diffusion to the subretinal space has not been proved in experimental studies, but also because some studies demonstrated the success of pneumatic displacement of subretinal blood without concomitant injection of r-TPA(90,91,93).
Therefore, Haupert introduced a hybrid technique in 2001, combining the concepts of pneumatic displacement with subretinal r-Tpa(94).
After a few changes, this technique is currently used in some centers, as described: pars plana vitrectomy, removal of the posterior hyaloid, injection of r-TPA (12.5 or 25 μg/0.1 mL) into the subretinal clot using a 39 or 41-gauge flexible translocation cannula and fluid-air exchange followed by prone position(95).
The advantages of this technique over the surgical blood removal are less intra and post-operative complications, which is probably due to the smaller extent of tissue manipulation and consequent reduction in retinal injury(83,94,95). Usually after successful blood displacement, the patient experience a short gain in visual acuity that frequently drop down because of recurrent and massive haemorrhage or progression of the underlying disease. Such cases might benefit from more invasive procedures as vitrectomy and 180º retinectomy for direct access to subretinal space allowing the complete removal of the underlying blood and CNV(96,97).
Since the advent of anti-VEGF therapy, a combined intraocular injection of bevacizumab and r-TPA seems to be a promising treatment for AMD with submacular haemorrhage. Whatever the chosen procedure, close observation and additional intra- and/or postoperative anti-VEGF treatments might potentially prevent CNV related complications(98-100). n selected cases, namely patients with multiple diseases and little subretinal blood, monotherapy with intravitreal anti-VEGF could offer some chance of visual improvement with a minimally invasive approach(101).
More recently, there has been a trend towards more complex associated procedures: vitrectomy and gas endotamponade with subretinal injection of r-TPA and anti-VEGF agents (1.25 mg bevacizumab). The thinking is that vitrectomy may relieve any vitreomacular traction, preventing wet-AMD from getting worse. Regarding subretinal anti-VEGF application, it ensures the direct delivery of the drug at the site of the CNV, which may enhance its effect; the short half-life of r-TPA (around 5 minutes) may reduce the risk of drugs interaction when given alongside anti-VEGF agents, particularly when injected into the confined subretinal space(98,102-104). In 2013, Martel et al. advocated a combined subretinal injection of r-TPA, bevacizumab and filtered air (0,2 mL), followed by intravitreal fluid-air or hexafluoride gas exchange and up-right positioning in order to get an easier hemorrhage displacement due to the lowering haemorrhage’s buoyancy obtained by the subretinal air(105).
As occurs in other macular diseases and also previous referred, the detection on OCT of an ellipsoid layer and external limiting membrane line beneath the fovea are strongly correlated with good visual prognosis(106). A study that used SD-OCT to evaluate the preoperative features of submacular haemorrhage and PED in nAMD, found out that the presence of an intact ellipsoid layer and lower height of submacular haemorrhage were predictors of good postoperative BCVA. In contrast, the preoperative characteristics of PED had no effects on visual prognosis(107). In the recent Stanesc-Segall literature review the greatest improvement is obtained when a combined approach with vitrectomy, subretinal r-TPA, intravitreal gas and anti-VEGF is performed(104). However, according to others, subretinal r-TPA, intravitreal gas and anti-VEGF treatment without vitrectomy, which is a less invasive procedure, had also good functional results with acceptable safety and fewer complications(100).
Concomitant systemic anticoagulation or platelet anti-aggregant medication should be discontinued if possible, especially in monocular patients. With the improvement of vitreoretinal surgery and instrumentation, described techniques are reasonably effective, although with widely variable outcome, depending on the severity of the underlying disease. Post-surgical addition of anti-VEGF medications appears to help preserve vision over time.
Regardless treatment modality, the benefits of surgery diminish with an increasing duration, extension and thickness of submacular haemorrhage(108).
Submacular haemorrhage associated with CNV due to AMD is a potentially visually devastating condition and to date, there is no consensus regarding an optimal treatment.
References - Surgery in AMD
References - Surgery in AMD
- Macular Photocoagulation Study Group. Laser photocoagulation of subfoveal neovascular lesions in age-related macular degeneration. Results of a randomized clinical trial. Arch Ophthalmol 1991;109(9):1220-1231.
- Macular Photocoagulation Study Group. Laser photocoagulation of subfoveal recurrent neovascular lesions in age-related macular degeneration. Results of a randomized clinical trial. Arch Ophthalmol 1991;109(9):1232-1241.
- Macular Photocoagulation Study Group. Laser photocoagulation of subfoveal neovascular lesions of age-related macular degeneration. Updated findings from two clinical trials. Arch Ophthalmol 1993;111(9):1200-1209.
- Krebs I, Brannath W, Glittenberg C, Zeiler F, Sebag J, Binder S. Posterior vitreomacular adhesion: a potential risk factor for exudative age-related macular degeneration? Am J Ophthalmol 2007;144(5):741-746.
- Schulze S, Hoerle S, Mennel S, Kroll P. Vitreomacular traction and exudative age-related macular degeneration. Acta Ophthalmol 2008;86(5):470-481.
- Lee SJ, Lee CS, Koh HJ. Posterior vitreomacular adhesion and risk of exudative age-related macular degeneration: paired eye study. Am J Ophthalmol 2009;147(4):621-626.
- Mojana F, Cheng L, Bartsch DU, Silva GA, Kozak I, Nigam N, Freeman WR. The role of abnormal vitreomacular adhesion in age-related macular degeneration: spectral optical coherence tomography and surgical results. Am J Ophthalmol 2008;146(2):218-227.
- Foulds WS. Factors influencing visual recovery in retinal detachment surgery. Trans Ophthalmol Soc U K 1980;100(Pt 1):72-77.
- Machemer R, Steinhorst UH. Retinal separation, retinotomy, and macular relocation: I. Experimental studies in the rabbit eye. Graefes Arch Clin Exp Ophthalmol 1993;231(11):629-634.
- Machemer R, Steinhorst UH. Retinal separation, retinotomy, and macular relocation: II. A surgical approach for age-related macular degeneration? Graefes Arch Clin Exp Ophthalmol 1993;231(11):635-641.
- Machemer R. Macular translocation. Am J Ophthalmol 1998;125(5):698-700.
- Wolf S, Lappas A, Weinberger AW, Kirchhof B. Macular translocation for surgical management of subfoveal choroidal neovascularizations in patients with AMD: first results. Graefes Arch Clin Exp Ophthalmol 1999;237(1):51-57.
- Eckardt C, Eckardt U, Conrad HG. Macular rotation with and without counter-rotation of the globe in patients with age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol 1999;237(4):313-325.
- Ninomiya Y, Lewis JM, Hasegawa T, Tano Y. Retinotomy and foveal translocation for surgical management of subfoveal choroidal neovascular membranes. Am J Ophthalmol 1996;122(5):613-621.
- Toth CA, Machemer R. Macular translocation. In: Berger JW, Fine SL, Maguire MG, eds. Age-related macular degeneration. Saint-Louis, USA. Mosby. 1999;353-362.
- Holgado S, Enyedi LB, Toth CA, Freedman SF. Extraocular muscle surgery for extorsion after macular translocation surgery new surgical technique and clinical management. Ophthalmology 2006;113(1):63-69.
- Pertile G, Claes C. Macular translocation with 360 degree retinotomy for management of age-related macular degeneration with subfoveal choroidal neovascularization. Am J Ophthalmol 2002;134(4):560-565.
- Aisenbrey S, Bartz-Schmidt KU, Walter P, Hilgers RD, Ayertey H, Szurman P, Thumann G. Long-term follow-up of macular translocation with 360 degrees retinotomy for exudative age-related macular degeneration. Arch Ophthalmol 2007;125(10):1367-1372.
- van Romunde SHM, Polito A, Bertazzi L, Guerriero M, Pertile G.. Long-term results of full macular translocation for choriodal neovascularization in age-related macular degeneration. Ophthalmology 2015;122(7):1366-1374.
- Chen FK, Patel PJ, Uppal GS, Tufail A, Coffey PJ, Da Cruz L.. Long-term outcomes following full macular translocation surgery in neovascular age-related macular degeneration. Br J Ophthalmol 2010;94(10):1337-1343.
- American Academy of Ophthalmology. Macular translocation. Ophthalmology 2000;107(5):1015-1018.
- Charles S, Calzada J, Wood B. Submacular surgery and macular translocation. In: Charles S, Calzada J, Wood B, eds. Vitreous microsurgery. Fourth Edition. Philadelphia, USA. Lippincott Williams & Wilkins. 2007;14:163-171.
- Mruthyunjaya P, Stinnett SS, Toth CA. Change in visual function after macular translocation with 360 degrees retinectomy for neovascular age-related macular degeneration. Ophthalmology 2004;111(9):1715-1724.
- Toth CA, Lapolice DJ, Banks AD, Stinnett SS. Improvement in near visual function after macular translocation surgery with 360-degree peripheral retinectomy. Graefes Arch Clin Exp Ophthalmol 2004;242(7):541-548.
- Wong D, Stanga P, Briggs M, Lenfestey P, Lancaster E, Li KK, Lim KS, Groenewald C. Case selection in macular relocation surgery for age related macular degeneration. Br J Ophthalmol 2004;88(2):186-190.
- Uppal G, Milliken A, Lee J, Acheson J, Hykin P, Tufail A, da CL. New algorithm for assessing patient suitability for macular translocation surgery. Clin Experiment Ophthalmol 2007;35(5):448-457.
- Aisenbrey S, Lafaut BA, Szurman P, Grisanti S, Luke C, Krott R, Thumann G, Fricke J, Neugebauer A, Hilgers RD, Esser P, Walter P, Bartz-Schmidt KU. Macular translocation with 360 degrees retinotomy for exudative age-related macular degeneration. Arch Ophthalmol 2002;120(4):451-459.
- Vugler A, Carr AJ, Lawrence J, Chen LL, Burrell K, Wright A, Lundh P, Semo M, Ahmado A, Gias C, da Cruz L, Moore H, Andrews P, Walsh J, Coffey P. Elucidating the phenomenon of HESC-derived RPE: anatomy of cell genesis, expansion and retinal transplantation. Exp Neurol 2008;214(2):347-61.
- Terasaki H, Ishikawa K, Suzuki T, Nakamura M, Miyake K, Miyake Y. Morphologic and angiographic assessment of the macula after macular translocation surgery with 360 degrees retinotomy. Ophthalmology 2003;110(12):2403-8.
- De Juan E Jr, Loewenstein A, Bressler NM, Alexander J. Translocation of the retina for management of subfoveal choroidal neovascularization II: a preliminary report in humans. Am J Ophthalmol 1998;125(5):635-646.
- Pieramici DJ, de Juan E Jr, Fujii GY, Reynolds SM, Melia M, Humayun MS, Schachat AP, Hartranft CD. Limited inferior macular translocation for the treatment of subfoveal choroidal neovascularization secondary to age-related macular degeneration. Am J Ophthalmol 2000;130(4):419-428.
- Fujii GY, de Juan E Jr, Pieramici DJ, Humayun MS, Phillips S, Reynolds SM, Melia M, Schachat AP. Inferior limited macular translocation for subfoveal choroidal neovascularization secondary to age-related macular degeneration: 1-year visual outcome and recurrence report. Am J Ophthalmol 2002;134(1):69-74.
- Fujii GY, Pieramici DJ, Humayun MS, Schachat AP, Reynolds SM, Melia M, de Juan E Jr. Complications associated with limited macular translocation. Am J Ophthalmol 2000;130(6):751-762.
- Eandi CM, Giansanti F, Virgili G. Macular translocation for neovascular age-related macular degeneration. Cochrane Database Syst Rev 2008;(4):CD 006928.
- De Juan E Jr, Machemer R. Vitreous surgery for hemorrhagic and fibrous complications of age-related macular degeneration. Am J Ophthalmol 1988;105(1):25-29.
- Lambert HM, Capone A Jr, Aaberg TM, Sternberg P Jr, Mandell BA, Lopez PF. Surgical excision of subfoveal neovascular membranes in age-related macular degeneration. Am J Ophthalmol 1992;113(3):257-262.
- Berger AS, Kaplan HJ. Clinical experience with the surgical removal of subfoveal neovascular membranes. Short-term postoperative results. Ophthalmology 1992;99(6):969-975.
- Thomas MA, Grand MG, Williams DF, Lee CM, Pesin SR, Lowe MA. Surgical management of subfoveal choroidal neovascularization. Ophthalmology 1992;99(6):952-968.
- Bressler NM. Submacular surgery. Are randomized trials necessary? Arch Ophthalmol 1995;113(12):1557-1560.
- Bressler NM, Bressler SB, Hawkins BS, Marsh MJ, Sternberg P Jr, Thomas MA. Submacular surgery trials randomized pilot trial of laser photocoagulation versus surgery for recurrent choroidal neovascularization secondary to age-related macular degeneration: I. Ophthalmic outcomes submacular surgery trials pilot study report number 1. Am J Ophthalmol 2000;130(4):387-407.
- Conti SM, Kertes PJ. Surgical management of age-related macular degeneration. Can J Ophthalmol 2005;40(3):341-351.
- Ibanez HE, Williams DF, Thomas MA, Ruby AJ, Meredith TA, Boniuk I, Grand MG. Surgical management of submacular hemorrhage. A series of 47 consecutive cases. Arch Ophthalmol 1995;113(1):62-69.
- Wade EC, Flynn HW Jr, Olsen KR, Blumenkranz MS, Nicholson DH. Subretinal hemorrhage management by pars plana vitrectomy and internal drainage. Arch Ophthalmol 1990;108(7):973-978.
- Lewis H. Intraoperative fibrinolysis of submacular hemorrhage with tissue plasminogen activator and surgical drainage. Am J Ophthalmol 1994;118(5):559-568.
- Grossniklaus HE, Gass JD. Clinicopathologic correlations of surgically excised type 1 and type 2 submacular choroidal neovascular membranes. Am J Ophthalmol 1998;126(1):59-69.
- Grossniklaus HE, Green WR. Histopathologic and ultrastructural findings of surgically excised choroidal neovascularization. Submacular Surgery Trials Research Group. Arch Ophthalmol 1998;116(6):745-749.
- Soloman S, Dong LM, Haller JA, Gilson MM, Hawkins BS, Bressler NM; SST research group and the SST adverse event review committee. Risk factors for rhegmatogenous retinal detachment in the submacualr surgery trials. Retina 2009;29(6):819-824.
- Hawkins BS, Bressler NM, Miskala PH, Bressler SB, Holekamp NM, Marsh MJ, Redford M, Schwartz SD, Sternberg P Jr, Thomas MA, Wilson DJ. Surgery for subfoveal choroidal neovascularization in age-related macular degeneration: ophthalmic findings: SST report no. 11. Ophthalmology 2004;111(11):1967-1980.
- Miskala PH, Bass EB, Bressler NM, Childs AL, Hawkins BS, Mangione CM, Marsh MJ. Surgery for subfoveal choroidal neovascularization in age-related macular degeneration: quality-of-life findings: SST report no. 12. Ophthalmology 2004;111(11):1981-1992.
- Bressler NM, Bressler SB, Childs AL, Haller JA, Hawkins BS, Lewis H, MacCumber MW, Marsh MJ, Redford M, Sternberg P Jr, Thomas MA, Williams GA. Surgery for hemorrhagic choroidal neovascular lesions of age-related macular degeneration: ophthalmic findings: SST report no. 13. Ophthalmology 2004;111(11):1993-2006.
- Childs AL, Bressler NM, Bass EB, Hawkins BS, Mangione CM, Marsh MJ, Miskala PH. Surgery for hemorrhagic choroidal neovascular lesions of age-related macular degeneration: quality-of-life findings: SST report no. 14. Ophthalmology 2004;111(11):2007-2014.
- Thumann G, Aisenbrey S, Schraermeyer U, Lafaut B, Esser P, Walter P, Bartz-Schmidt KU. Transplantation of autologous iris pigment epithelium after removal of choroidal neovascular membranes. Arch Ophthalmol 2000;118(10):1350-1355.
- Binder S, Stolba U, Krebs I, Kellner L, Jahn C, Feichtinger H, Povelka M, Frohner U, Kruger A, Hilgers RD, Krugluger W. Transplantation of autologous retinal pigment epithelium in eyes with foveal neovascularization resulting from age-related macular degeneration: a pilot study. Am J Ophthalmol 2002;133(2):215-225.
- Van Meurs JC, ter AE, Hofland LJ, van Hagen PM, Mooy CM, Baarsma GS, Kuijpers RW, Boks T, Stalmans P. Autologous peripheral retinal pigment epithelium translocation in patients with subfoveal neovascular membranes. Br J Ophthalmol 2004;88(1):110-113.
- Bindewald A, Roth F, Van MJ, Holz FG. Transplantation von retinalem Pigmentepithel (RPE) nach CNV-Exzision bei altersabhängiger Makuladegeneration. Techniken, Ergebnisse und Perspektiven. Ophthalmologe 2004;101(9):886-894.
- Aisenbrey S, Lafaut BA, Szurman P, Hilgers RD, Esser P, Walter P, Bartz-Schmidt KU, Thumann G. Iris pigment epithelial translocation in the treatment of exudative macular degeneration: a 3-year follow-up. Arch Ophthalmol 2006;124(2):183-188.
- Binder S, Krebs I, Hilgers RD, Abri A, Stolba U, Assadoulina A, Kellner L, Stanzel BV, Jahn C, Feichtinger H. Outcome of transplantation of autologous retinal pigment epithelium in age-related macular degeneration: a prospective trial. Invest Ophthalmol Vis Sci 2004;45(11):4151-4160.
- Peyman GA, Blinder KJ, Paris CL, Alturki W, Nelson NC Jr, Desai U. A technique for retinal pigment epithelium to extensive subfoveal scarring. Ophthalmic Surg 1991;22(2):102-108.
- Algvere PV, Berglin L, Gouras P, Sheng Y. Transplantation of fetal retinal pigment epithelium in age-related macular degeneration with subfoveal neovascularization. Graefes Arch Clin Exp Ophthalmol 1994;232(12):707–716.
- Algvere PV, Berglin L, Gouras P, Sheng Y, Kopp ED. Transplantation of RPE in age-related macular degenera- tion: Observations in disciform lesions and dry RPE atrophy. Graefes Arch Clin Exp Ophthalmol 1997;235(3):149–158.
- Stanga PE, Kychenthal A, Fitzke FW, Halfyard AS, Chan R, Bird AC, Aylward GW. Retinal pigment epithelium translocation after choroidal neovascular membrane removal in age-related macular degeneration. Ophthalmology 2002;109(8):1492-1498.
- MacLaren RE, Bird AC, Sathia PJ, Aylward GW. Long-term results of submacular surgery combined with macular translocation of the retinal pigment epithelium in neovascular age-related macular degeneration. Ophthalmology 2005;112(12):2081-2087.
- van Meurs JC, Van Den Biesen PR. Autologous retinal pigment epithelium and choroid translocation in patients with exudative age-related macular degeneration: short-term follow-up. Am J Ophthalmol 2003;136(4):688-695.
- Van Zeeburg EJ, Maaijwee KJ, Missotten TO, Heimann H, van Meurs JC. A free retinal pigment epithelium-choroid graft in patients with exudative age-related macular degeneration: Results up to 7 years. Am J Ophthalmol. 2012;153(1):120e7.e2.
- MacLaren RE, Uppal GS, Balaggan KS, Tufail AD, Ali RR, Aylward GW, DaCruz L. RPE Patch Graft Auto-Transplantation in Macular Degeneration: A Prospective Cohort Study. ARVO. Fort Lauderdale, USA, Apr 30-May 4, 2006. Invest Ophthalmol Vis Sci 2006; 47(13):2693.
- Joussen AM, Heussen FM, Joeres S, Llacer H, Prinz B, Rohrschneider K, Maaijwee KJ, Van MJ, Kirchhof B. Autologous translocation of the choroid and retinal pigment epithelium in age-related macular degeneration. Am J Ophthalmol 2006;142(1):17-30.
- Joussen AM, Joeres S, Fawzy N, Heussen FM, Llacer H, van Meurs JC, Kirchhof B. Autologous translocation of the choroid and retinal pigment epithelium in patients with geographic atrophy. Ophthalmology 2007;114(3):551-560.
- MacLaren RE, Uppal GS, Balaggan KS, Tufail A, Munro PM, Milliken AB, Ali RR, Rubin GS, Aylward GW, da CL. Autologous transplantation of the retinal pigment epithelium and choroid in the treatment of neovascular age-related macular degeneration. Ophthalmology 2007;114(3):561-570.
- Treumer F, Bunse A, Klatt C, Roider J. Autologous retinal pigment epithelium-choroid sheet transplantation in age related macular degeneration: morphological and functional results. Br J Ophthalmol 2007;91(3):349-353.
- Treumer F, Klatt C, Roider J. Autologe RPE-Chorioidea-Translokation bei exsudativer AMD. Eine Falldemonstration 10 konsekutiver Patienten. Ophthalmologe 2007;104(9):795-802.
- Maaijwee K, Heimann H, Missotten T, Mulder P, Joussen A, Van MJ. Retinal pigment epithelium and choroid translocation in patients with exudative age-related macular degeneration: long-term results. Graefes Arch Clin Exp Ophthalmol 2007;245(11):1681-1689.
- Maaijwee KJ, van Meurs JC, Kirchhof B, Mooij CM, Fischer JH, Mackiewicz J, Kobuch K, Joussen AM. Histological evidence for revascularisation of an autologous retinal pigment epithelium--choroid graft in the pig. Br J Ophthalmol 2007;91(4):546-550.
- Parolini, B, Alkabes, M, Baldi, A, Pinackatt, S. Visual recovery after autologous retinal pigment epithelium and choroidal patch in a patient with choroidal neovascularization secondary to angioid streaks: long-term results. Retin Cases Brief Rep. 2016 Fall;10(4):368-72.
- Maaijwee K, Van Den Biesen PR, Missotten T, Van Meurs JC. Angiographic evidence for revascularization of an rpe–choroid graft in patients with age-related macular degeneration. Retina 2008;28(3):498–503.
- Cereda MG, Parolini B, Bellesini E, Pertile G. Surgery for CNV and autologous choroidal RPE patch transplantation: exposing the submacular space. Graefes Arch Clin Exp Opththalmol 2010;248:37–47.
- Converse JM, Rapaport FT. The vascularisation of skin autografts and homografts. Ann Surg 1956;120:306–316.
- Bennett SR, Folk JC, Blodi CF, Klugman M. Factors prognostic of visual outcome in patients with subretinal hemorrhage. Am J Ophthalmol 1990;109(1):33-37.
- Berrocal MH, Lewis ML, Flynn HW Jr. Variations in the clinical course of submacular hemorrhage. Am J Ophthalmol 1996;122(4):486-493.
- Glatt H, Machemer R. Experimental subretinal hemorrhage in rabbits. Am J Ophthalmol 1982;94(6):762-773.
- Toth CA, Morse LS, Hjelmeland LM, Landers MB, III. Fibrin directs early retinal damage after experimental subretinal hemorrhage. Arch Ophthalmol 1991;109(5):723-729.
- Lewis H, Resuick SC, Flannery JG, Straatsma BR. Tissue plasminogen activator treatment of experimental subretinal hemorrhage. Am J Ophthalmol 1991;111:197-204.
- Peyman GA, Nelson NCJ, Alturki W, Blinder KJ, Paris CL, Desai UR, Harper CA 3rd. Tissue plasminogen activating factor assisted removal of subretinal hemorrhage. Ophthalmic Surg 1991;22(10):575-82.
- Kamei M, Tano Y. Tissue plasminogen activator-assisted vitrectomy: surgical drainage of submacular hemorrhage. Dev Ophthalmol 2009;44:82–88.
- Herriot WJ. Further experience in management of submacular hemorrhage with intravitreal tPA. Proceedings of the 1997 Update on Macular Surgery, American Academy of Ophthalmology. San Francisco, USA, October. 1997;82-84.
- Hesse L, Schmidt J, Kroll P. Management of acute submacular hemorrhage using recombinant tissue plasminogen activator and gas. Graefes Arch Clin Exp Ophthalmol 1999;237(4):273-277.
- Karlsson E, Carlsson J, Crafoord S, Jemt M, Martensson PA, Stenkula S. Tissue Plasminogen Activator and Expanding Gas Intravitreally in Treatment of Submacular Hemorrhage. Transactions of the Swedish Society of Ophthalmology 1997. Annual Meeting. Sundsvall, Sweden, August 27-30, 1997. Acta Ophthalmol Scand 1999;77(1): 119.
- Hassan AS, Johnson MW, Schneiderman TE, Regillo CD, Tornambe PE, Poliner LS, Blodi BA, Elner SG. Management of submacular hemorrhage with intravitreous tissue plasminogen activator injection and pneumatic displacement. Ophthalmology 1999;106(10):1900-1906.
- Handwerger BA, Blodi BA, Chandra SR, Olsen TW, Stevens TS. Treatment of submacular hemorrhage with low-dose intravitreal tissue plasminogen activator injection and pneumatic displacement. Arch Ophthalmol 2001;119(1):28-32.
- Hattenbach LO, Klais C, Koch FH, Gumbel HO. Intravitreous injection of tissue plasminogen activator and gas in the treatment of submacular hemorrhage under various conditions. Ophthalmology 2001;108(8):1485-1492.
- Ohji M, Saito Y, Hayashi A, Lewis JM, Tano Y. Pneumatic displacement of subretinal hemorrhage without tissue plasminogen activator. Arch Ophthalmol 1998;116(10):1326-1332.
- Daneshvar H, Kertes PJ, Leonard BC, Peyman GA. Management of submacular hemorrhage with intravitreal sulfur hexafluoride: a pilot study. Can J Ophthalmol 1999;34(7):385-388.
- Silva SR, Bindra MS. Early treatment of acute submacular haemorrhage secondary to wet AMD using intravitreal tissue plasminogen activator, C3F8, and an anti-VEGF agent. Eye (Lond) 2016;30(7):952-7.
- Kamei M, Misono K, Lewis H. A study of the ability of tissue plasminogen activator to diffuse into the subretinal space after intravitreal injection in rabbits. Am J Ophthalmol 1999;128(6):739-746.
- Haupert CL, McCuen BW, Jaffe GJ, Steuer ER, Cox TA, Toth CA, Fekrat S, Postel EA. Pars plana vitrectomy, subretinal injection of tissue plasminogen activator, and fluid-gas exchange for displacement of thick submacular hemorrhage in age-related macular degeneration. Am J Ophthalmol 2001;131(2):208-215.
- Olivier S, Chow DR, Packo KH, MacCumber MW, Awh CC. Subretinal recombinant tissue plasminogen activator injection and pneumatic displacement of thick submacular hemorrhage in Age-Related macular degeneration. Ophthalmology 2004; 111 (6): 1201-1208.
- Thompson JT, Sjaarda RN. Vitrectomy for the treatment of submacular hemorrhages from macular degeneration: a comparison of submacular hemorrhage/membrane removal and submacular tissue plasminogen activator-assisted pneumatic displacement. Trans Am Ophthalmol Soc. 2005;103:98-107, discussion 107.
- Fine HF, Iranmanesh R, Del Priore LV, Barile GR, Chang LK, Chang S, Schiff WM. Surgical outcomes after massive subretinal hemorrhage secondary to age-related macular degeneration. Retina. 2010;30(10):1588-94.
- Treumer F, Klatt C, Roider J, Hillenkamp J. Subretinal co-application of recombinant tissue plasminogen activator and bevacizumab for neovascular age-related macular degeneration with submacular hemorrhage. Br J Ophthalmol 2010;94(1):48-53.
- Kamei M, Tano Y, Maeno T, Ikuno Y, Mitsuda H, Yuasa T. Surgical removal of submacular hemorrhage using tissue plasminogen activator and perfluorocarbon liquid. Am J Ophthalmol. 1996;121(3):267-75.
- Van Zeeburg EJT, van Meurs JC. Literature review of recombinant tissue plasminogen activator used for recent-onset submacular haemorrhage displacement in age-related macular degeneration. Ophthalmologica 2013;229(1):1-14.
- Kim JH, Chang YS, Kim JW, Kim CG, Yoo SJ, Cho HJ.. Intravitreal anti–vascular endothelial growth factor for submacular hemorrhage from choroidal neovascularization. Ophthalmology 2014;121(4):926–935.
- Simpson AR, Petrarca R, Jackson TL. Vitreomacular adhesion and neovascular age-related macular degeneration. Surv Ophthalmol. 2012;57(6):498-509.
- Treumer F, Roider J, Hillenkamp J. Long-term outcome of subretinal co-application of recombinant tissue plasminogen activator and bevacizumab followed by repeated intravitreal anti-VEGF injections for neovascular age-related macular degeneration with submacular hemorrhage. Br J Ophthalmol 2012;96(5):708-713.
- Stanescu-Segall D, Balta F, Jackson TL. Submacular hemorrhage in neovascular age-related macular degeneration: A synthesis of the literature. Surv Ophthalmol 2016;61(1):18-32.
- Martel JN, Mahmoud TH. Subretinal pneumatic displacement of subretinal hemorrhage. JAMA Ophthalmol 2013;131(12):1632-5.
- Ueda-Arakawa N, Tsujikawa A, Yamashiro K, Ooto S, Tamura H, Yoshimura N. Visual prognosis of eyes with submacular hemorrhage associated with exudative age-related macular degeneration. Jpn J Ophthalmol 2012;56(6): 589–598.
- Hirashima T, Moriya T, Bun T, Utsumi T, Hirose M, Oh H. Optical coherence tomography findings and surgical outcomes of tissue plasminogen activator-assisted vitrectomy for submacular hemorrhage secondary to age-related macular degeneration. Retina 2015;35(10):1969–78.
- Kitagawa Y, Shimada H, Mori R, Tanaka K, Yuzawa M.Intravitreal Tissue Plasminogen Activator, Ranibizumab, and Gas Injection for Submacular Hemorrhage in Polypoidal Choroidal Vasculopathy. Ophthalmology 2016; 123(6):1278-86.